Kenny CW CHAN
The first report of AIDS described 5 patients with Pneumocystis carinii pneumonia. All of them had laboratory-confirmed mucosal infection with Candida albicans. This led the authors to suggest the 'possibility of a cellular-immune dysfunction related to a common exposure that predisposes individuals to opportunistic infections such as pneumocystosis and candidiasis'.1 Indeed candidiasis is now known to be the most common opportunistic fungal infection in HIV disease.
In HIV-positive patients, both colonisation and disease in oral cavity and other mucosal surfaces is common, especially with C. albicans, at rates that increase with progression of disease.2 Such HIV-induced alterations as decreased salivary IgA levels, impairment of mucosal CD4 cells, altered cytokine secretion and a shift to Th2 cytokine expression in saliva likely contribute to Candida colonisation and disease.3 Candidiasis is also associated with use of oral contraceptives, smoking, dentures, and immune suppression related to chemotherapeutic agents, corticosteroids, pregnancy and diabetes. In HIV infection, these conditions may aggravate or precipitate candidiasis.
There are two main forms of candidiasis - mucosal and disseminated. As with other opportunistic infections, the introduction of highly active antiretroviral therapy (HAART) has led to a decreased incidence of oral and oesophageal candidiasis.4
Mucosal candidiasis, in the form of oral, oesophageal or vaginal infections, is common in HIV/AIDS. The incidence of oral candidiasis in HIV infection varies from 7% to 93%, depending on patient mix, diagnostic criteria and study methods. A review of local HIV-infected women showed that the prevalence of oral candidiasis was 9% and that of vaginal candidiasis 28%.5 In an early study of HIV-infected women, Candida vaginitis was the commonest HIV-associated clinical condition in women.6 Infections tend to persist and are associated with a lower CD4 count and higher HIV viral load.7 In the revised 1993 CDC case definition,8 vaginal candidiasis that was persistent, frequent or poorly responsive to therapy, as well as oral candidiasis, became a designated HIV-associated category B condition. In recent years, however, the status of recurrent vaginal candidiasis as a sentinel of HIV infection has been called into question, as it has never been supported by prospective controlled studies. In the current classification, oesophageal candidiasis is an AIDS-defining condition, occurring with a CD4<200/μL and usually in conjunction with oral candidiasis. Rarely, oral candidiasis may occur in the setting of primary HIV infection when the CD4 count is significantly reduced.
Unlike mucosal candidiasis, disseminated candidiasis is remarkably uncommon in AIDS. This is likely due to relatively adequate neutrophil function in most HIV-infected individuals. Although candidaemia has been reported in AIDS, it usually occurred in the presence of other risk factors such as neutropaenia, parenteral nutrition, abdominal surgery, broad spectrum antibiotics, cancer and corticosteroid use.9,10
Oral candidiasis is an independent prognostic factor of HIV disease progression,11 and it therefore could predict the occurrence of Pneumocystis jiroveci pneumonia (PCP). Its presence is an indication for PCP prophylaxis and consideration of treatment with HAART.
The symptoms of oral thrush are burning pain, altered taste sensation, and difficulty with swallowing. These contribute to poor oral intake and weight loss. However, most patients are asymptomatic. There are 4 forms of disease on examination:
Pseudomembranous candidiasis (thrush) is characterised by the presence of white or creamy plaques on the dorsal surface of the tongue, the hard or soft palate and buccal mucosa. Such lesions can be scraped off with a tongue depressor, leaving a raw or bleeding surface.
Erythematous candidiasis appears on the dorsal surface of the tongue as a patchy depilated area, sometimes with corresponding (kissing) smooth red patches on the palate. The patient is more likely to be symptomatic with complaints of burning pain while eating spicy foods.
Angular cheilitis appears as cracking, fissuring, or erythema at the corner of the mouth.
Occasionally, Candida causes hyperkeratosis (candidal leukoplakia), causing diagnostic difficulty with oral hairy leukoplakia. Diagnosis can be confirmed by biopsy.
C. albicans is a common commensal of the skin and mucosal surface. Its isolation therefore does not equate disease. Diagnosis of candidiasis is usually made on the grounds of clinical symptoms and physical examination. It may be possible to scrape off thrush. In atypical cases, microscopic examination of a KOH smear helps support the diagnosis by showing pseudohyphae and blastospores. Culture is not routinely indicated, but if performed, it identifies the Candida species and helps predict resistance. In vitro resistance test may be considered in recurrent and refractory infections.
The major differential diagnosis of oral candidiasis is oral hairy leukoplakia, which is usually located at the sides of the tongue and cannot be scraped off. Candidal leukoplakia cannot be wiped off either but will regress with prolonged antifungal therapy. Biopsy will also show yeasts.
Fluconazole (100 mg qd for 7-14 d or till thrush resolves; some authorities recommend a loading dose of 200 mg on Day 1), with its superior effectiveness in HIV infected patients is the standard of care and is particularly indicated in the event of low CD4 or recurrent disease. The other azoles: Itraconazole oral solution at 200 mg qd is as effective as fluconazole but is less well tolerated. Ketoconazole and itraconazole capsules are less effective and not recommended.
Clotrimazole tablet (qid to 5x/d for 7-14 d) and nystatin oral suspension (5 mL swish and swallow qid to 5x/day for 7-14 d) are cheaper but lesser alternatives. They are justified in patients with relatively high CD4 count and less severe disease.
Oral candidiasis typically responds well to fluconazole, but recurs unless there is significant immune recovery with HAART. Recurrences after treatment may be due to C. albicans, a new strain of C. albicans with resistance, or with a non-albicans species of Candida such as C. glabrata, C. parapsilosis, C. krusei and C. dublinensis. The latter may be confused with C. albicans as both are associated with germ tube formation in the laboratory. It is not uncommon that multiple species or more than one genotype of C. albicans is isolated. Species other than C. albicans are generally less susceptible to therapy, and arise mainly with low CD4 count and after repeated or prolonged antifungal treatment.
Recurrent disease is usually managed in the same way as initial disease. The disease may become refractory to treatment, in which case treatment failure has occurred and diagnosis should be confirmed with microscopy or culture. Treatment options are:
If culture has identified non-albicans species or if in vitro sensitivity results are available, they should be taken into account in the selection of treatment. With immune recovery after the use of HAART, previously refractory oral candidiasis generally becomes more responsive to treatment.
In a similar fashion to oral thrush, oesophageal candidiasis may be presumptively diagnosed when typical symptoms occur, especially in the presence of oral candidiasis. Typical symptoms are retrosternal pain, odynophagia and dysphagia. Constitutional symptoms, including fever, occur only occasionally.
Upper endoscopy is warranted in case of treatment failure to identify adherent, whitish mucosal plaques and superficial mucosal ulcerations, and to differentiate from such lesions as herpetic oesophagitis, CMV oesophagitis, aphthous ulceration, AZT-induced ulceration, and idiopathic or HIV oesophagitis. Biopsy of the lesions typically shows characteristic Candida yeast forms in tissue, accompanied by a neutrophilic response. The species can be identified by culture. Occasionally, oesophageal candidiasis is asymptomatic and diagnosed when upper endoscopy is performed for an unrelated cause.
Oesophageal candidiasis may uncommonly be complicated by bezoar formation, perforation and fistula formation. If there is severe neutropaenia, oesophagitis may also lead to candidaemia or bacteraemia.
Systemic therapy is necessary for oesophageal candidiasis in the form of fluconazole 200-400 mg qd for 2-3 weeks. An acceptable alternative is itraconazole oral solution 100-200 mg bid. Itraconazole capsules and ketoconazole are less effective.
Failure of symptoms to respond to fluconazole in 7-10 days should preferably be followed by upper endoscopy to confirm the diagnosis and rule out coexisting diseases, after which the following options may be used:
The Candida species involved may provide valuable guidance to the choice of therapy. Resistance testing is not routinely performed and may be difficult to interpret. The natural history of oesophageal candidiasis is recurrence within two to three months after treatment. Long term suppressive therapy (secondary prophylaxis) with fluconazole (100-200 mg qd) or itraconazole solution (200 mg qd) may be considered in frequently recurrent disease. It is important that HAART be initiated upon the diagnosis of oesophageal candidiasis.
Patients may be asymptomatic or may complain of perivaginal pruritus or dysuria. A gynaecologic examination typically shows erythematous labia, shallow, linear ulcerations on the introitus and/or satellite papules beyond the main area of erythema. Candida discharge is classically thick, white and adherent.
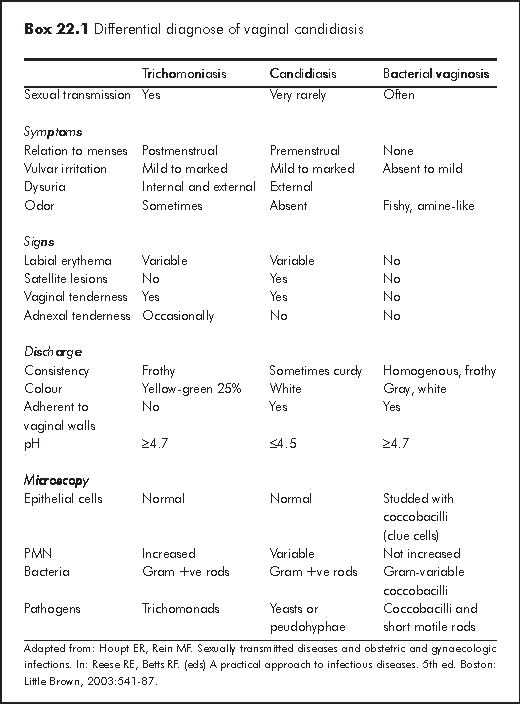
Diagnosis of vaginal candidiasis is often made clinically and treatment begun empirically. Other symptoms include external dysuria, vaginal soreness and vulvar burning. With atypical presentation or treatment failure, a 10% KOH smear should be performed, for demonstrating budding yeast and pseudohyphae. Gram stain and culture are also useful. Vaginal pH is normal, i.e. <=4.5. The whiff test should also be negative in vaginal candidiasis. Note that isolation of Candida in an asymptomatic woman does not indicate treatment, as Candida may also exist as commensal in the vagina. The following table summarises the distinguishing features of vaginal candidiasis from trichomoniasis and bacterial vaginosis, the major differential diagnoses (Box 22.1).

Topical therapy generally suffices. The following regimens, as used in HIV-negative women, are appropriate as first line therapy. They are also safe in pregnancy.
Single dose therapy
3-day therapy
7-day azole therapy
14-day therapy
Although 7-day therapy is more effective than the more abbreviated regimens and nystatin, the difference is probably not clinically significant. Aggravating factors should be ruled out and controlled if possible. These include diabetes, douching, pregnancy, corticosteroid use, concurrent antibiotic, hormonal contraceptives, tight-fitting synthetic underwear and possibly spermicides.
Recurrent infection can be a major problem in both HIV positive and negative women. It is important that the presence of aggravating factors be reviewed and controlled if there is any. On ruling out other causes of chronic vaginitis, one may re-treat with the above regimens but with a longer duration. Alternatively one may consider the following oral regimens:
Culture is useful in identifying non-albicans Candida spp. With oral azoles, one must be assured that the patient is receiving adequate contraception.
Suppressive therapy with intermittent fluconazole is generally not advised but may be considered in the case of recurrent vaginal candidiasis, defined as >=4 episodes of symptomatic vaginal candidiasis annually. The diagnosis should be confirmed by culture before such treatment. The sexual partner may also be examined for balanitis and treated, though its value in decreasing the occurrence of vaginal candidiasis is controversial. The options of suppressive therapy are:
With these regimens, cost is a major concern. Long term use of the cheaper ketoconazole is not advisable because of likelihood of interaction with protease inhibitors and unreliable absorption in AIDS patients. The need for such maintenance treatment is reevaluated after 6 months. Although suppressive therapy is usually effective, 30-40% of women have recurrent disease once therapy is discontinued.
Claims for certain probiotics, such as Lactobacilli, to prevent vaginal candidiasis and bacterial vaginosis are unsubstantiated. They are not recommended for use, especially in advanced immunosuppression.
As a strategy to prevent candidiasis, the prevention of exposure is impractical as Candida is common in the environment and as a commensal. The use of fluconazole is effective in prospective controlled trials to prevent cryptococcosis as well as mucosal candidiasis in advanced HIV disease. However, mortality is not reduced. Prolonged primary prophylaxis is therefore not advised, "because of the effectiveness of therapy for acute disease, the low mortality associated with mucosal candidiasis, the potential for resistant Candida organisms to develop, the possibility of drug interactions, and the cost of prophylaxis".12
As secondary prophylaxis, chronic treatment with fluconazole 100-200 mg daily may be considered in frequently recurrent esophageal candidiasis. However, recurrent oesophagitis is becoming uncommon in the era of HAART.
The mainstay of treatment of candidiasis is the use of antifungals. Since oral and vaginal candidiasis can occur in the presence of a relatively high CD4 count, the impact of HAART on their occurrence may be less than that on oesophageal candidiasis. Nevertheless, a higher CD4 count and a more competent CD4 repertoire should theoretically contribute to a more rapid cure with antifungals and possibly less resistance. It is not uncommon to observe that HAART therapy results in resolution of refractory, drug-resistant mucosal candidiasis, even with limited immune recovery.13 It is hypothesised that HIV protease inhibitors have specific anti-Candida efficacy by inhibiting the secreted aspartic proteinases of Candida albicans, a key virulence factor.14,15
In Hong Kong, the readily available antifungals appropriate for mucosal candidiasis include:
The azoles: isoconazole (Gyno-Travogen) vaginal tablet, clotrimazole (Gyne-Lotremin) vaginal tablet, tioconazole (Gyno-Trosyd) vaginal tablet, cream and ointment, and oral ketoconazole, itraconazole, and fluconazole
The polyenes: nystatin (suspension 105u/mL and pessary), amphotericin B (IV and lozenge), and talsutin (coformulated tetracycline and amphotericin B) vaginal tablet
Use of topical agents is limited to oral and vaginal candidiasis. Systemic agents e.g. fluconazole, are indicated for second line therapy, advanced AIDS and oesophageal candidiasis. Among the systemic azoles, efficacy is generally highest with fluconazole and itraconazole solution. Ketoconazole is inferior, probably because of impaired absorption in advanced AIDS. The relative inefficacy of clotrimazole troches and nystatin suspension, however, is partially offset by their low cost. It is emphasised that for oesophageal candidiasis, only systemic agents are useful. Flucytosine may be used only in combination with an azole or amphotericin B for severe disease, since resistance rapidly emerges if flucytosine is used alone.
The newer antifungals of voriconazole, caspofungin and micafungin have been found to be effective in treatment of oesophageal candidiasis. Although they are not readily available, they should be considered in refractory disease.
In mucosal candidiasis refractory to azoles and amphotericin B, anecdotal success has been reported of treatment with interferon-γ and recombinant granulocyte-macrophage colony stimulating factor. However, with the extensive antifungal armamentarium now available, there is limited role for their use.
Despite years of use, resistance to amphotericin B is still relatively rare. On the other hand, widespread use of systemic azoles in recent years has led to the emergence of primary and secondary resistance, especially in advanced AIDS.16 Although a standard for antifungal sensitivity testing against Candida has been defined and continues to be refined by the National Committee for Clinical Laboratory Standards (NCCLS), its use in the clinical setting is still very limited. Furthermore, not all azole-resistant candidiasis can be explained by in vitro resistance. This implies that in the event of failure, treatment is usually changed without clear objective criteria. In general the options are increasing the dosage, changing to a new drug, or using combinations of drugs. Commonly, resistance is caused by infection with non-albicans Candida which are intrinsically less susceptible to treatment.
Systemic azoles are metabolised by the cytochrome P450 (CYP) enzymes and hence may have significant drug interactions. Their metabolism is increased by rifamycins, long-acting barbiturates and phenytoin. Fluconazole has the least propensity to cause drug interactions. Voriconazole, ketoconazole and itraconzole are all contraindicated with cisapride, terfenadine and astemizole for fear of ventricular arrhythmia. They may also increase the levels of sulfonyureas, HMG Co-A reductase inhibitors (statins) omeprazoles and ergot alkaloids. There is also interaction with protease inhibitors which are also metabolised by the same enzymes. In particular, full-dose ritonavir (400 mg bid or more) is contraindicated with voriconazole. Baby dose of ritonavir (200 mg bid or less) as pharmacokinetic enhancer may be used with caution. Efavirenz is also contraindicated as it decreases levels of voriconazole by 70%. Fluconazole may increase the level of rifabutin leading to uveitis.
Caspofungin is much less prone to serious drug interactions. Nevertheless, with concurrent efavirenz, nevirapine, rifampin and phenytoin, caspofungin should be increased to 70 mg/d.
Craniofacial and skeletal abnormalities have been reported following prolonged in utero exposure to fluconazole. Itraconazole is embryotoxic in animal systems. Consequently systemic azoles are contraindicated in pregnancy. Teratogenic effects on rats have also been observed with flucytosine which is therefore also contraindicated in pregnancy.
Centers for Disease Control (CDC). Pneumocystis pneumonia--Los Angeles. MMWR Morb Mortal Wkly Rep 1981;30:250-2.
Vargas KG, Joly S. Carriage frequency, intensity of carriage, and strains of oral yeast species vary in the progression to oral candidiasis in human immunodeficiency virus-positive individuals. J Clin Microbiol 2002;40:341-50.
Challacombe SJ, Sweet SP. Oral mucosal immunity and HIV infection: current status. Oral Dis 2002;8 Suppl 2:55-62.
Mocroft A, Oancea C, van Lunzen J, et al. Decline in esophageal candidiasis and use of antimycotics in European patients with HIV. Am J Gastroenterol 2005;100:1446-54.
Chan KCW, Wong KH, Lee SS. HIV Infection in Women: the Hong Kong Experience to date. First Annual Scientific Meeting, Hong Kong Society for Infectious Disease [Abstract]. Hong Kong, 1997
Carpenter CC, Mayer KH, Fisher A, Desai MB, Durand L. Natural history of acquired immunodeficiency syndrome in women in Rhode Island. Am J Med 1989;86(6 Pt 2):771-5.
Duerr A, Heilig CM, Meikle SF, et al. Incident and persistent vulvovaginal candidiasis among human immunodeficiency virus-infected women: Risk factors and severity. Obstet Gynecol 2003;101:548-56.
1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Recomm Rep 1992;41(RR-17):1-19.
Manfredi R, Calza L, Chiodo F. Dual Candida albicans and Cryptococcus neoformans fungaemia in an AIDS presenter: a unique disease association in the highly active antiretroviral therapy (HAART) era. J Med Microbiol 2002;51:1135-7.
Garbino J, Kolarova L, Lew D, Hirschel B, Rohner P. Fungemia in HIV-infected patients: a 12-year study in a tertiary care hospital. AIDS Patient Care STDS 2001;15:407-10.
Katz MH, Greenspan D, Westenhouse J, et al.Progression to AIDS in HIV-infected homosexual and bisexual men with hairy leukoplakia and oral candidiasis. AIDS 1992;6:95-100.
Kaplan JE, Masur H, Holmes KK; USPHS; Infectious Disease Society of America. Guidelines for preventing opportunistic infections among HIV-infected persons--2002. Recommendations of the U.S. Public Health Service and the Infectious Diseases Society of America. MMWR Recomm Rep 2002;51(RR-8):1-52.
Zingman BS. Resolution of refractory AIDS-related mucosal candidiasis after initiation of didanosine plus saquinavir. N Engl J Med 1996;334:1674-5.
Bektic J, Lell CP, Fuchs A, et al. HIV protease inhibitors attenuate adherence of Candida albicans to epithelial cells in vitro. FEMS Immunol Med Microbiol 2001;31:65-71.
Cassone A, Tacconelli E, De Bernardis F, et al. Antiretroviral therapy with protease inhibitors has an early, immune reconstitution-independent beneficial effect on Candida virulence and oral candidiasis in human immunodeficiency virus-infected subjects. J Infect Dis 2002;185:188-95.
Darouiche RO. Oropharyngeal and esophageal candidiasis in immunocompromised patients: treatment issues. Clin Infect Dis 1998;26:259-74.