
Nelson LS LEE, Ian CT TSE
Herpes simplex virus (HSV) is a DNA virus, belonging to the family Herpesviridae. Sequence homology between HSV types 1 and 2 is about 50%. Serologic assays of antibody can differentiate between the two by using type specific antigens, such as the gG1 and gG2 proteins. Upon primary infection, the virus establishes latency in neuronal cells of ganglia. Reactivation is frequent especially in the first year after infection. The mechanism of reactivation may be through alteration in viral proteins that are responsible for maintaining latency. UV light, trauma, immunosuppression and stress have been cited as factors causing reactivation. As cell-mediated immunity is essential to the containment of infection, it is understandable why HSV infection is particularly important in HIV disease.
Varicella zoster virus (VZV) is also a herpesvirus. As such it shares structural characteristics with HSV. Furthermore, the virus causes similar histopathologic features, e.g. intranuclear inclusions and multinucleated giant cells. VZV causes two major syndromes: varicella (chickenpox) and zoster (shingles).
Traditionally, HSV-1 has been known to be associated with orolabial herpes and HSV-2 with genital herpes. However, investigations have amply documented the increased trend of genital HSV-1 in many parts of the world. In a recent report, the proportion of HSV-1 anogenital isolates rose from <10% in the early 1980s to around 35% in the late 1990s and early 2000s.1
HSV-1 is commonly acquired in childhood, particularly in poorer socio-economic conditions. Seroprevalence of HSV-1 rises rapidly with age. By the age of 24, the seroprevalence of HSV-1 in Hong Kong approaches 80-90% [unpublished data; Dr. J Lo, Government Virus Unit].
HSV-2 is less prevalent than HSV-1. Using western blot method, the seroprevalence of HSV-2 in Hong Kong's general population (>=25 years) was found to be 17-18%.2,3 The seroprevalence has conceivably increased among STD clinic attendees and commercial sex workers (Box 25.1). HSV-2 sexual transmission rate between discordant couples is about 10-15% per year.4

As a sexually transmitted infection, primary herpes may serve as an indicator of high risk sexual behaviour. From the pathological point of view, HSV-2 infection affects HIV and vice versa:
(a) Effect of HSV-2 on HIV
There is a 2-4 times increase in the risk of HIV acquisition in those with genital herpes. HIV is consistently found in herpetic ulcers; these mucosal or epithelial breaks therefore may serve as portal of exit or entry for HIV. Concomitant HSV infection also up-regulates HIV replication, resulting in their higher plasma and genital tract levels. HIV-infected partner who reported genital lesions has 4-folded risk in transmitting HIV to un-infected partners, adjusted for the viral load.5-7
(b) Effect of HIV on HSV-2
Patients with HIV may have more frequent, severe and prolonged episodes of recurrences of genital herpes especially in those with a low CD4 count <200/μL. There is a higher rate of subclinical shedding of HSV-2 (i.e. in the absence of obvious genital lesions) in those HIV-infected. HIV also increases the risk of acquisition and transmission of HSV-2. HSV-2 infection actually defines AIDS in the case of chronic ulcers of more than one month duration, bronchitis, pneumonitis or oesophagitis.8-11
The hallmark of HSV infection is the development of multiple small painful vesicles on an erythematous base involving a mucocutaneous site. The vesicles ulcerate and then heal by re-epithelialisation. HSV-1 commonly causes orolabial lesions while HSV-2 infects primarily the genital and perinanal regions. Clinical manifestations depend on the site of inoculation, degree of immunosuppression, subtype of HSV and whether the infection is the primary infection or reactivation disease. The incubation period ranges from 2 to 14 days (median 7 days). Reactivation disease is generally of shorter duration and consists of milder symptoms, but both primary and reactivation infections are often clinically asymptomatic. It is becoming clear that most people continue to shed HSV during periods when there are no signs or symptoms of disease. Indeed, most HSV transmission occurs during these asymptomatic periods or from a partner who does not realise that he or she is infected.
Genital herpes classically presents as grouped vesicles in the genitalia. These lesions subsequently become pustules then erosions, thereafter crusting and healing within 2-3 weeks. Tender inguinal lymphadenopathy and systemic symptoms such as fever, headache, malaise, myalgias and meningismus are more common in primary infection than the reactivation disease. In women, the primary genital infection frequently results in cervicitis, thus partially explaining the high frequency of perinatal transmission when herpes is acquired late in pregnancy.
HSV pharyngitis is not uncommon in those with primary genital herpes probably because of orogenital contact. Ulcers are seen in the pharnynx which are covered by a white exudate. These are accompanied by tender cervical lymphadenopathy, and a vesicular eruption on the lips, tongue and buccal mucosa. It may occur more commonly in those with HIV infection. HSV-1 gingivostomatitis and pharyngitis usually occur alone without genital involvement after transmission by kissing or other intimate contact. Reactivation in the form of herpes labialis was reported, and may occur at any CD4 count. Chronic and severe disease may occur if the CD4 count is below 100/μL.
Proctitis caused by HSV has been described primarily in HIV-infected male patients. It usually presents with fever, pruritus, rectal pain, tenesmus and rectal discharge. Urinary retention, impotence and sacral paraesthesia may be present particularly in primary HSV infection. Sigmoidoscopy often demonstrates ulceration. Concomitant Neisseria gonorrhoeae infection has to be excluded.
HSV keratitis is characterised by dendritic corneal ulcerations. It is probably a result of autoinoculation. Recurrences are common. Progressive scarring may ultimately lead to blindness. In disseminated HSV, chorioretinitis may also occur. Either HSV or varicella zoster virus may cause an acute retinal necrosis syndrome which is difficult to treat.
Upon initial genital infection, the herpesvirus lies dormant in sacral ganglia. Reactivation follows unidentified stimuli. Immune suppression increases the rate and severity of reactivation. HSV-2 also tends to reactivate more commonly than HSV-1. In general, systemic symptoms are absent and lesions are confined to the genitalia. Prodromal symptoms of tingling sensation or pain are common before a recurrence. In HIV infection, defective cell mediated immunity increases the chance of complications. Recurrences and subclinical shedding of virus become more common. Furthermore, the following manifestations may occur in either primary infection or reactivation (usually at a lower CD4 count):
(a) A persistent mucocutaneous form of genital herpes with extensive involvement of the genital and perianal area.
(b) HSV oesophagitis: This AIDS-defining complication is usually seen in patients with CD4 cell count less than 50/μL. The classic symptom is odynophagia or burning retrosternal pain. Concomitant orolabial ulcerations are common. As the symptoms are clinically indistinguishable from oesophagitis caused by Candida species or cytomegalovirus, definitive diagnosis requires endoscopically obtained biopsy. Most herpetic lesions are seen in the distal third of the oesophagus. Rare complications include oesophageal strictures and perforation.
(c) Disseminated HSV infections (hepatitis, pneumonia, or encephalitis): These disseminated forms of HSV infection are rare. Unlike in other form of immunosuppression where HSV-1 encephalitis usually localised in temporal lobe, the HSV encephalitis in HIV-infected individual often has diverse areas of brain involvement, including brain stem.
Varicella is a common childhood infection in Hong Kong. Most adults and adolescents with HIV infection are already infected with the virus.
Thus the major manifestations are those related to zoster. During primary varicella infection, VZV enters cutaneous endings of sensory nerves and migrates to dorsal root ganglia where it establishes latency in a similar fashion to HSV. Reactivation in the form of zoster occurs at all ages in the immunocompetent, but is more common after 50 and in the immunocompromised. HIV disease predisposes towards zoster that is more severe, extensive, and common.
Typical zoster presents as painful vesiculopustular eruption distributed along a dermatome. Radicular pain may precede the rash by 48 to 72 h. Resolution by crusting and re-epithelialisation may be followed by scarring. There is no evidence that postherpetic neuralgia is more common in HIV disease. HIV-associated zoster can also be unusually severe, involving more than one dermatome.12 In general, zoster occurs at an earlier age than the immunocompetent, reflecting the younger HIV infected age group. Furthermore, most herpes zoster infection first develops when CD4 count drops to 300-500/μL.13 The zoster may also recur, as an immune reconstitution syndrome, after initiation of antiretroviral agents in severely immunocompromised patients when their CD4 count rises to a similar level.
Chronic disseminated zoster may present as verrucous or ulcerative lesions. A less common presentation is a persistent localised form of zoster. Both forms are typically resistant to treatment with acyclovir. Zoster involving the ophthalmic branch of the trigeminal nerve (zoster ophthalmicus) may result in keratitis and uveitis and may be more common in HIV disease. VZV can also cause acute retinal necrosis which carries a poor prognosis and which mandates aggressive antiviral treatment.14 Apart from cutaneous dissemination, visceral dissemination may also occur, resulting in life threatening infections of the liver, lungs and CNS.
Diagnosis of oral and genital herpes is often based on clinical grounds, although the classical presentations can be absent. Genital herpes is also one differential diagnosis of the so-called genital ulcer adenopathy syndrome. The other diagnoses are syphilis, chancroid, lymphogranuloma venereum (LGV), donovanosis or giant idiopathic aphthous genital ulcers that may rarely occur in HIV disease. However, genital herpes remains the commonest cause of genital ulcer adenopathy syndrome in Hong Kong. HSV infection can be detected by the following tests:
(a) Viral culture. Isolation of HSV in cell culture is the preferred virologic test for patients who seek medical treatment for genital ulcers or other mucocutaneous lesions. However, the sensitivity of culture is low, especially for recurrent lesions, and declines rapidly as lesions begin to heal. Specimens are most likely to yield virus if they are taken from the early lesions within the first one or two days after their formation. Viral culture isolates should be typed to determine if HSV-1 or HSV-2 is the cause of the infection. Lack of HSV detection does not indicate a lack of HSV infection, as viral shedding is intermittent.
(b) Tzanck smear. The smear is obtained by scraping the base of a vesicle with a scalpel. The material is then fixed in alcohol and stained with Wright or Giemsa stain. Multinucleated giant cells will be seen as with other herpesvirus infections. However, it is only positive in about 50% of cases.
(c) Direct immunofluorescence study with monoclonal antibodies (DFA) on samples obtained from active lesions, and PCR assay are useful tests. PCR assay for HSV DNA is a highly sensitive and specific. Detection of HSV DNA in CSF is particularly helpful in the early diagnosis of HSV encephalitis. However these tests may be limited by their cost and availability.
(d) Type-specific serologic tests for HSV: Type-specific HSV serologic assays might be useful in the following scenarios: (1) recurrent genital symptoms or atypical symptoms with negative HSV cultures; (2) a clinical diagnosis of genital herpes without laboratory confirmation; and (3) a partner with genital herpes. Some specialists believe that HSV serologic testing should be included in a comprehensive evaluation for STDs among persons with multiple sex partners, HIV infection, and among MSM at increased risk for HIV acquisition. The sensitivities of the FDA-approved glycoprotein G type-specific tests for the detection of HSV-2 antibody vary from 80-98%, and false-negative results might be more frequent at early stages of infection. The specificities of these assays are >96%.
Diagnosis of typical herpes zoster is often a clinical one. Atypical or disseminated forms of zoster may require laboratory confirmation by viral culture, Tzanck smear or DFA. PCR may be utilised for the diagnosis of CNS infection or pneumonitis.
Prompt recognition of infection allows antivirals to be started early. They help control the symptoms and signs of herpes episodes and reduce viral shedding. In typical presentations, treatment should not be delayed by waiting for laboratory confirmation. Patient-initiated treatment upon first sign of recurrence is effective. Counselling regarding the natural history of genital herpes, sexual and perinatal transmission, and methods to reduce transmission is integral to clinical management.15 Algorithm 25(A) (end of chapter) is constructed to illustrate the approach to the management of genital herpes.
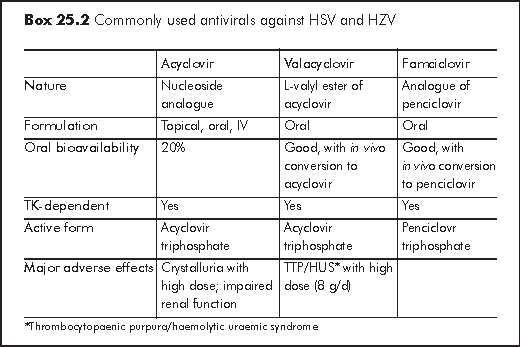
Drugs against HSV and varicella zoster (VZV) infections act by inhibiting viral DNA polymerase (Box 25.2). Randomised trials have determined the clinical benefits of three antiviral medications: acyclovir, valacyclovir, and famciclovir. Acyclovir is the "prototype" drug. Valacyclovir is the valine ester of acyclovir and has enhanced absorption after oral administration. Similarly, famciclovir also has high oral bioavailability. All three drugs have been demonstrated to reduce the severity and frequency of clinical and subclinical reactivations, and to reduce viral shedding in HIV-infected patients. While acyclovir and valacyclovir have been shown to reduce plasma HIV RNA levels; this has not been shown for famciclovir. Ganciclovir is equally effective in treatment of HSV, but it is too toxic for routine use. All the three available formulations of acyclovir, topical, oral and intravenous, are generally well tolerated; however dosage adjustment is needed in patients with renal insufficiency, especially during high dose therapy.15
Topical acyclovir - Although 5% acyclovir ointment slightly shortens the duration of symptoms in primary genital infection, it has now fallen out of favour because it (a) does not work in reactivations, (b) does not alleviate systemic symptoms, (c) has to be applied 6 times a day, and (d) care must be taken to avoid autoinoculation.
Oral acyclovir - This is the treatment of choice in most situations. Early treatment is more effective. In first episode genital herpes, acyclovir is given as 200 mg 5x/day or 400 mg tid for 7-10 days. In recurrent disease, acyclovir is given as 200 mg 5x/day or 400 mg tid for 5 days (effective episodic treatment of recurrent herpes requires initiation of therapy within 1 day of lesion onset or during the prodrome). The patient should be provided with a supply of drug or a prescription for the medication with instructions to initiate treatment immediately when symptoms begin. To reduce recurrence, acyclovir 400 mg bid is recommended (Box 25.3).15 Herpes simplex infection among HIV-infected patients may be more severe and may respond less well to treatment. Therefore both the duration and dose will need to be increased if response is unsatisfactory.
Intravenous acyclovir - This is used for serious infections such as disseminated infection, pneumonitis, hepatitis or CNS infections. Alternatively, it may be considered in suspected resistance and in patients with poor absorption or drug compliance. The standard recommended dose is 5-10 mg/kg IV q8h for 5-7 d or until clinical resolution is attained, and followed by oral therapy if appropriate to complete at least 10 days of treatment. The higher end of the range, i.e. 10 mg/kg, is mandatory for CNS infections as CSF level is usually only 30-50% of that in plasma.

Acyclovir, valacyclovir, famciclovir and ganciclovir require viral thymidine kinase (TK) for antiviral action. The most common mechanism of HSV resistance is mediated through altered substrate specificity of the enzyme. Resistance is rare in immunocompetent hosts, but in HIV disease about 5% of HSV infections were resistant to acyclovir in the pre-HAART era. Acyclovir resistance is less commonly observed nowadays with the use of HAART. If HSV resistance is suspected, a viral isolate should be obtained for sensitivity testing. All acyclovir-resistant strains are resistant to valacyclovir, and the majority are also resistant to famciclovir. As foscarnet dose not require phoshporylation for its activity, it is frequently effective for treatment of acyclovir-resistant genital herpes. Foscarnet is given at 40 mg/kg body weight IV every 8 hours until clinical resolution is attained. Topical cidofovir gel 1% applied to the lesions once daily for 5 consecutive days has also been tried with success, but commercial preparation is not available yet.
Acute management of zoster lesions involves pain management with analgesics, local treatment and specific antivirals.
In general higher doses of antivirals are required for zoster than HSV infections and should be started within 72 hours of rash onset. Similar to treatment of HSV, antivirals for VZV reduce viral shedding and duration of disease. They may also reduce the risk of postherpetic neuralgia. Acyclovir, valacyclovir and famciclovir are the preferred first line drugs. Treatment is continued for 7-10 days or until lesions have crusted. Although no comparative trials have been performed, acyclovir is considered inferior because of its relatively poor bioavailability and more frequent dosing.
The comparative doses of the commonly used antivirals against zoster are listed in Box 25.4. Most HIV infected patients with localised zoster require only oral treatment as an outpatient. However, cutaneous dissemination and visceral involvement require intravenous acyclovir for treatment.

Resistant zoster, generally presenting as persistent lesions refractory to treatment, is usually due to deficient or altered thymidine kinase. Cross resistance among the first line antivirals is expected. In this case, IV foscarnet at 40 mg/kg q8h may be effective.
Most patients have been infected by varicella prior to HIV. In the rare occurrence of varicella in an HIV infected person, IV acyclovir treatment should be considered as visceral dissemination is not uncommon. Susceptible patients without history of chickenpox and antibody should be given varicella zoster immune globulin (VZIG) as prophylaxis within 96 hours of exposure. Note that VZIG is ineffective as treatment. As a live attenuated vaccine, the varicella vaccine is contraindicated in HIV patients.
It is important to advise patient on the use of barrier contraceptives and the knowledge that shedding of virus is often subclinical. Sex should be abstained when there are recognisable recurrences. The use of condom reduces but does not eliminate the risk of infection as some genital lesions may not be covered by the condom. The use of daily suppressive therapy in frequent relapsers (>=6 recurrences per year) is beneficial not only to reduce the frequency of relapses (by 70-80%) but also reduce virus shedding,15 minimising transmission of both HSV and HIV. Both acyclovir and valacyclovir can be considered. Breakthrough recurrences do not necessarily reflect drug resistance, and higher doses of suppressive therapy may be attempted. However, this approach may involve a long period of antiviral treatment and the decision needs to be individualised. The frequency of recurrent genital herpes diminishes over time in many patients, and the patient's psychological adjustment to the disease might change. Therefore, periodically during suppressive treatment (e.g., once a year), providers should discuss the need to continue therapy with the patient. An alternative approach is to treat each recurrent episode to ameliorate the symptoms.
It has also been shown that while HAART does reduce the incidence of symptomatic genital herpes and its severity, it has a modest and not statistically significant effect on subclinical HSV-2 reactivation.16 Therefore HSV-2 shedding is still possible even when the immune system is reconstituted.
Genital herpes in pregnancy is an important issue.17 It is recommended that women with recurrent genital herpetic lesions at the onset of labour deliver by caesarean section to prevent neonatal herpes. However, caesarean section does not completely eliminate the risk of HSV transmission to the infant. The safety of systemic acyclovir, valacyclovir, and famciclovir therapy in pregnant women has not been definitively established. However, available data do not indicate an increased risk for major birth defects in women treated with these antiviral agents during the first trimester. Acyclovir may be administered orally to pregnant women with first episode genital herpes or severe recurrent herpes and should be administered IV to pregnant women with severe HSV infection. Acyclovir treatment late in pregnancy reduces the frequency of caesarean sections among women who have recurrent genital herpes by diminishing the frequency of recurrences at term, and many specialists recommend such treatment.18 No data support the use of antiviral therapy among HSV seropositive women without a history of genital herpes.
Haddow LJ, Dave B, Mindel A, et al. Increase in rates of herpes simplex virus type 1 as a cause of anogenital herpes in western Sydney, Australia, between 1979 and 2003. Sex Transm Infect 2006;82:255-9.
Hong Kong Department of Health. Epidemiology of herpes simplex virus type 2 in Hong Kong. Hong Kong STD/AIDS Update 1999;5:7-8.
Lo JY, Lim WW, Ho DW, Field PR, Cunningham AL. Difference in seroprevalence of herpes simplex virus type 2 infection among antenatal women in Hong Kong and southern China. Sex Transm Infect 1999;75:123.
Pattman R, Snow M, Handy P, editors, Oxford Handbook of Genitourinary Medicine, HIV and AIDS. Oxford: Oxford University Press, 2005; p 239.
Freeman EE, Weiss HA, Glynn JR, Cross PL, Whitworth JA, Hayes RJ. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS 2006;20:73-83.
Corey L, Wald A, Celum CL, Quinn TC. The effects of herpes simplex virus-2 on HIV-1 acquisition and transmission: a review of two overlapping epidemics. J Acquir Immune Defic Syndr 2004;35:435-45.
Moriuchi M, Moriuchi H, Williams R, Straus SE. Herpes simplex virus infection induces replication of human immunodeficiency virus type 1. Virology 2000;278:534-40.
Posavad CM, Wald A, Kuntz S, et al. Frequent reactivation of herpes simplex virus among HIV-1-infected patients treated with highly active antiretroviral therapy. J Infect Dis 2004;190:693-6.
Schacker T, Ryncarz AJ, Goddard J, Diem K, Shaughnessy M, Corey L. Frequent recovery of HIV-1 from genital herpes simplex virus lesions in HIV-1-infected men. JAMA 1998;280:61-6.
Augenbraun M, Feldman J, Chirgwin K, et al. Increased genital shedding of herpes simplex virus type 2 in HIV-seropositive women. Ann Intern Med 1995;123:845-7.
1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Recomm Rep 1992;41(RR-17):1-19.
Cohen PR, Beltrani VP, Grossman ME. Disseminated herpes zoster in patients with human immunodeficiency virus infection. Am J Med 1988;84:1076-80.
Friedman-Kien AE, Lafleur FL, Gendler E, et al. Herpes zoster: a possible early clinical sign for development of acquired immunodeficiency syndrome in high-risk individuals. J Am Acad Dermatol 1986;14:1023-8.
Hellinger WC, Bolling JP, Smith TF, Campbell RJ. Varicella-zoster virus retinitis in a patient with AIDS-related complex: case report and brief review of the acute retinal necrosis syndrome. Clin Infect Dis 1993;16:208-12.
US CDC. 2006. Sexually transmitted diseases treatment guidelines. MMWR 2006;55:RR-11.
Posavad CM, Wald A, Kuntz S, et al. Frequent reactivation of herpes simplex virus among HIV-1-infected patients treated with highly active antiretroviral therapy. J Infect Dis 2004;190:693-6.
Brown ZA, Wald A, Morrow RA, Selke S, Zeh J, Corey L. Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant. JAMA 2003;289:203-9.
Sheffield JS, Hill JB, Hollier LM, et al. Valacyclovir prophylaxis to prevent recurrent herpes at delivery: a randomised clinical trial. Obstet Gynecol 2006;108:141-7.