Vincent WS WONG, Henry LY CHAN
Owing to similar modes of transmission, human immunodeficiency virus (HIV) infected patients are not uncommonly co-infected with hepatitis B virus (HBV) and hepatitis C virus (HCV). Worldwide, however, epidemiology of HIV-HBV and HIV-HCV co-infections varies as a result of difference in background hepatitis infections and routes of HIV transmission. In Hong Kong, unlike western countries, concomitant HBV is more prevalent than HCV infection in HIV infected subjects.
In the era of highly active antiretroviral therapy (HAART), HIV-infected patients are living longer. Coupled with a reduction in conventional opportunistic infections, more patients with HBV-HIV or HCV-HIV co-infection develop liver cirrhosis and liver-related mortality. The impact of viral interaction on the natural history and treatment has become increasingly important in the clinical management of HBV-HIV and HCV-HIV co-infections. In this chapter, the latest scientific development regarding HBV-HIV and HCV-HIV co-infection is reviewed, with highlights of key issues in the management of these patients.
Approximately 350 million people worldwide are infected by HBV. In general, HBV is not directly cytopathic. Damage to hepatocytes is related to host immunological activity against HBV inside the hepatocytes. Among Asian patients who acquire the infection at infancy, the natural history of chronic HBV is divided into three phases. In the immune tolerance phase, there is little host immune response to HBV. This usually occurs during childhood and teenage. The liver enzymes are normal despite high HBV DNA level and positive hepatitis B e-antigen (HBeAg). The immune clearance phase occurs in early adulthood when host immunity starts to clear HBV-infected hepatocytes. This is reflected by elevated liver enzymes and fluctuating HBV DNA. Successful viral suppression is manifested as HBeAg-seroconversion and undetectable HBV DNA. These fortunate individuals would then enter the quiescent phase and have good overall prognosis. In patients who fail to achieve successful viral suppression, chronic or intermittent inflammation of the liver parenchyma may result in cirrhosis and possibly hepatocellular carcinoma.
In HIV-infected patients who are first exposed to HBV in adulthood, the clinical presentation is often more subtle and may be silent. Alanine aminotransferase (ALT) is usually lower and jaundice is infrequently seen. This reflects reduced immune response in these patients. Nevertheless, HIV positive individuals are less likely to clear the virus and more prone to develop into chronic infection. In a study in Australia, 23% of the HIV-infected patients developed chronic HBV infection after acute hepatitis B, compared to 4% of non-HIV-infected patients.1 The chance of seroconversion to anti-HBs was proportional to the CD4 count.
HIV-infected patients with chronic hepatitis B commonly have high viral load and positive HBeAg. Both markers have recently been shown to be associated with the development of hepatocellular carcinoma.2,3 In a multi-centre study involving 5293 homosexual men, liver-related mortality was 14.2 per 1000 person years in HBV-HIV co-infected cases, compared to 1.7 per 1000 in those infected with HIV alone and 0.8 per 1000 in those infected with HBV.4 On the other hand, occult HBV infection is more common, i.e. detectable HBV DNA but hepatitis B surface antigen (HBsAg) negative. The presence of isolated anti-hepatitis B core antibody (anti-HBc) but negative HBsAg and anti-hepatitis B surface antibody (anti-HBs) occurs in 16% of HIV-infected patients.5 This phenomenon may represent occult HBV infection, co-infection with HCV which suppresses HBsAg expression, divergent results to different anti-HBs assays, previous HBV infection or vaccination, and emergence of hepatitis B surface mutants. Patients with isolated positive anti-HBc should have HBV DNA and anti-HCV checked. Although traditionally anti-HBs signifies immunity against HBV, HBsAg and anti-HBs may co-exist in HIV-infected patients. This can be due to reactivation of latent HBV infection or active infection with another HBV serotype.
As for HCV, more than 170 million people are chronically infected by the virus. HCV-HIV co-infection affects 25% of HIV patients and 5-10% HCV patients in Europe and North America.6 This is attributed to similar routes of transmission of the two viruses - intravenous drug abuse and use of unscreened blood products. In Hong Kong, the occurrence is less common because of the relatively smaller proportion of HIV infection in drug users. HCV-HIV co-infected patients have worse liver-related outcomes than those infected with HCV only. After acute HCV infection, around 85% of the non-HIV-infected patients become chronically infected. By contrast, only 5-10% of HIV-infected individuals spontaneously clear HCV. In addition, HCV-HIV co-infected patients tend to have high HCV RNA, which is translated into increased risk of liver cirrhosis and hepatocellular carcinoma. The rate of liver disease progression is also inversely proportional to the CD4 count.7 On the other hand, there is no definite evidence that HCV infection alters the course of HIV infection.
At present, interferon-alpha, pegylated interferon-alpha (PEG-IFN-α), lamivudine and adefovir dipivoxil are registered for the treatment of chronic hepatitis B in Hong Kong. These agents have been extensively investigated, but literature on their use in HBV-HIV co-infected patients is relatively scarce.
Conventional interferon and PEG-IFN-α clear HBV via their immunomodulatory and antiviral actions. Conventional interferon is usually given three times per week as subcutaneous injections. Because HIV-infected patients have poor immunity, it is not surprising that their response to interferon is also suboptimal. In a randomised controlled trial involving 50 homosexual men, interferon-alfa treatment resulted in 24% overall treatment success defined as undetectable HBV DNA by branched chain assay and HBeAg seroconversion. However, the response was only 8.4% among the subgroup of patients with HBV-HIV co-infection. In another retrospective analysis of 141 HBeAg-positive patients, HBV-HIV co-infection was associated with poorer treatment response, higher rate of relapse and risk of cirrhosis.8 The difference was more prominent among patients with low CD4 count. By adding a polyethylene glycol side-chain to the interferon molecule, PEG-IFN alfa has longer half-life, allowing it to be given once a week. This also results in increased therapeutic efficacy.9 However, the side effect profile of the two different preparations is similar. The data on the use of PEG-IFN in patients with HBV-HIV co-infection are not available at present.
Oral nucleos(t)ide analogues suppress HBV replication by inhibiting the DNA polymerase. As HBV replication involves reverse transcription as in the case of HIV, a number of registered and investigational drugs for HBV treatment have been used in the treatment of HIV before. Lamivudine is given at a dose of 150 mg twice daily for HIV and 100 mg once daily for HBV treatment. In HBV-HIV co-infected patients, the early response to lamivudine is satisfactory. In a cohort of 40 patients with HBV-HIV co-infection, lamivudine treatment at 300 mg per day resulted in HBeAg seroconversion in 19%, a figure that was comparable to that of patients without HIV infection.10 The main problem of long-term lamivudine therapy is the almost inevitable emergence of drug-resistant mutants at the tyrosine-methionine-aspartate-aspartate (YMDD) motif of the HBV polymerase gene (YMDD mutants). In HBV-HIV co-infected patients receiving lamivudine, YMDD mutants emerge at a rate of 20% per year and reach 90% by 4 years.11 This mutation rate is higher than that in patients infected with HBV alone.
Adefovir dipivoxil is an effective treatment for YMDD mutants and can be used as salvage therapy for lamivudine failure. At 10 mg per day, the drug has no appreciable effect on HIV RNA or CD4 counts in HIV-infected patients. On the other hand, adefovir at 30 mg per day has some anti-HIV activity but may result in nephrotoxicity. Resistance to adefovir is quite rare (<1%) in the first year, but may rise to 25% by 5 years. Unfortunately, this drug only has modest activity and slow onset of action, especially when used in patients with HBV-HIV co-infection. A study of 29 HBV-HIV co-infected patients with YMDD mutants receiving adefovir for 144 weeks showed that only 25% had HBV DNA below 200 copies/mL and only 2 patients developed HBeAg seroconversion.12
Entecavir is a potent oral anti-HBV drug with no anti-HIV activity. In chronic hepatitis B patients without HIV infection, entecavir results in HBV DNA drop of 6 log copies in HBeAg positive cases and 5 log copies in HBeAg negative cases. Although lamivudine has some cross-resistance with entecavir, a higher dose (1 mg daily) of entecavir has been used successfully in patients with lamivudine resistance. In co-infected patients who do not need anti-HIV treatment, giving adefovir or entecavir as the first-line therapy may carry the advantage of reserving the other antiviral drugs with HBV as well as HIV activity. Nevertheless, different treatment strategies have not been tested head-to-head in HBV-HIV co-infected patients.
Tenofovir disoproxil fumarate has in vitro activity against both HBV and HIV. It is currently registered for the treatment of HIV at a dose of 300 mg per day but not for HBV infection. A retrospective analysis of 65 patients with HBV-HIV co-infection showed that tenofovir reduced HBV DNA by 4.6 log copies/mL in HBeAg-positive patients and 2.5 log copies/mL in HBeAg-negative patients.13 A non-randomised study also showed that tenofovir was superior to adefovir in patients with lamivudine-resistant HBV.14 After treatment for 48 weeks, 100% of patients receiving tenofovir versus 44% of patients receiving adefovir had HBV DNA below 105 copies/mL. Tenofovir resistance has not been reported up to 130 weeks of treatment.14 Currently, the combination of tenofovir and emtricitabine (or lamivudine) is a standard treatment for HBV-HIV co-infection when the treatment of both infections is indicated.
The standard treatment for chronic hepatitis C is PEG-IFN alfa plus ribavirin. Currently, there are two preparations of PEG-IFN alfa: PEG-IFN alfa-2a is given at 180 μg subcutaneously once a week, while PEG-IFN alfa-2b is given at 1.5 μg/kg body weight per week. Ribavirin is given orally at a dose of 800-1200 mg per day, depending on the body weight and HCV genotype. This regimen results in sustained virological response, defined as negative HCV RNA at least 6 months after cessation of therapy, in up to 50% of non-HIV-infected patients infected with HCV genotype 1 and around 80% of patients infected with genotypes 2 and 3. The main limitation of this treatment regime is the side effect profile. The commonest side effects include flu-like symptoms, bone marrow suppression, psychiatric manifestations like depression and anxiety, and autoimmune reactions including thyroiditis.
In 3 randomised controlled trials involving a total of 1613 HCV-HIV co-infected patients reported in 2004, PEG-IFN alfa-2a plus ribavirin for 48 weeks resulted in a sustained virological response in 27-40%.15-17 This was significantly higher than patients who received conventional interferon and ribavirin (12-20%) or PEG-IFN alone (20%). Genotype 1, prior injection drug use and detectable HIV RNA at baseline were independent factors associated with poor treatment response. One of the major determinants of treatment success is however drug adherence, and treatment outcomes do not appear to be adversely affected in drug users who can adhere to treatment.
Current data do not suggest that chronic hepatitis B or C affect the natural course of HIV infection. However, hepatotoxicity secondary to HIV treatment is more common among patients with chronic viral hepatitis, especially among females with high baseline CD4 counts. The incidence of hepatotoxicity secondary to HAART has been reported to be between 2-18% among the major series.18,19 Invariably, HBV or HCV is among the factors associated with drug-induced liver injury. Regular monitoring of liver function tests is required during HAART.
Furthermore, a number of drug interactions are noteworthy. For the treatment of HCV-HIV co-infection, ribavirin should not be used together with nucleoside reverse transcriptase inhibitors didanosine and stavudine because of the risk of mitochondrial toxicity. This may lead to acute pancreatitis, lactic acidosis, hepatic steatosis and liver failure. Zidovudine should be avoided during HCV treatment because it increases the risk of anaemia and neutropaenia. Subgroup analysis of one study showed that protease inhibitors may reduce the treatment efficacy of PEG-IFN and ribavirin in HCV-HIV co-infected patients,17 but this needs further confirmation.
Regarding HBV-HIV co-infection, tenofovir and didanosine may lead to pancreatitis when used together. Tenofovir decreases the concentration of atazanavir, while atazanavir increases the concentration of tenofovir. Lopinavir-ritonavir increases the area under curve of tenofovir. Furthermore, adefovir decreases the concentration of saquinavir. These drug interactions can be managed with or without dosage adjustment.
Since HBV, HCV and HIV share common routes of transmission, initial assessment of an HIV infected patient should include a detailed history and physical examination looking for symptoms and signs of viral hepatitis and chronic liver disease. Liver function tests should be routinely checked together with viral serology including IgG anti-hepatitis A virus, HBsAg, anti-HBc and anti-HCV. Occult HBV and/or HCV infection is not uncommon in HIV-infected patients. If a patient has negative hepatitis serology but unexplained impairment in liver function, HBV DNA and HCV RNA should be checked as well.
In patients confirmed to have HBV-HIV or HCV-HIV co-infection, further assessment is necessary because the risk of advanced liver disease is increased. Complete blood count should be performed to look for thrombocytopaenia, and ultrasound scan should be arranged to rule out cirrhosis. Liver biopsy should be considered when a decision of treatment needs to be based on the severity of liver fibrosis. This is particularly important for genotype 1 HCV infection which is less likely associated with sustained virological response. Alpha-fetal protein should be performed every 6 months to screen for hepatocellular carcinoma. In patients confirmed to have liver cirrhosis, upper gastrointestinal endoscopy to screen for varices is recommended.
HIV patients should consider hepatitis A and B vaccination if IgG anti-hepatitis A virus, HBsAg and anti-HBs are negative. The response to HBV vaccine is dependent on the CD4 count. Thus, vaccination may be delayed in patients receiving HAART until immune reconstitution occurs. Anti-HBs antibody should be checked 4 weeks after the last dose of HBV vaccine. If anti-HBs is less than 10 IU/l, a second course or a booster dose can be given. For those who fail to seroconvert even after booster vaccine, HBsAg and anti-HBc should be checked yearly.
The treatment of co-infected patients is complicated. This should be done with a multi-disciplinary approach. Close liaison between hepatologists and physicians specialised in HIV is essential. For patients without immediate need for anti-HIV treatment, the decision on treatment is based on the HBV status. In general, the decision depends on the HBV DNA level and the disease severity. Treatment is considered if HBV DNA is >20,000 IU/mL in HBeAg-positive patients, >2,000 IU/mL in HBeAg negative patients and >200 IU/mL in cirrhotic patients. Among patients with high HBV DNA, the presence of liver inflammation and stage of fibrosis can be assessed by liver biopsy. Treatment is indicated if there is moderate to severe inflammation and/or evidence of fibrosis septa.
After a patient is confirmed to have active and/or advanced liver disease and requires HBV treatment, the choice of drugs is guided by the CD4 count and need of HIV treatment. If the CD4 count is high and HIV treatment not indicated, conventional interferon, PEG-IFN alfa, adefovir or entecavir can be considered. The other oral nucleos(t)ide analogues of lamivudine, emtricitabine or tenofovir can be reserved for their activity against HIV. Initiation of these nucleos(t)ide analogues without HAART is not recommended because a resistant strain of HIV will develop rapidly.
For patients with immediate need for HIV treatment, HAART consisting lamivudine/emtricitabine + tenofovir should be given. These agents with anti-HBV activity should not be the only drug in the regimen, to avoid development of HBV-resistant mutants.20 Entecavir is an option in such situation. Interferon-based treatment is rarely indicated for patients with established liver cirrhosis due to poor tolerability.
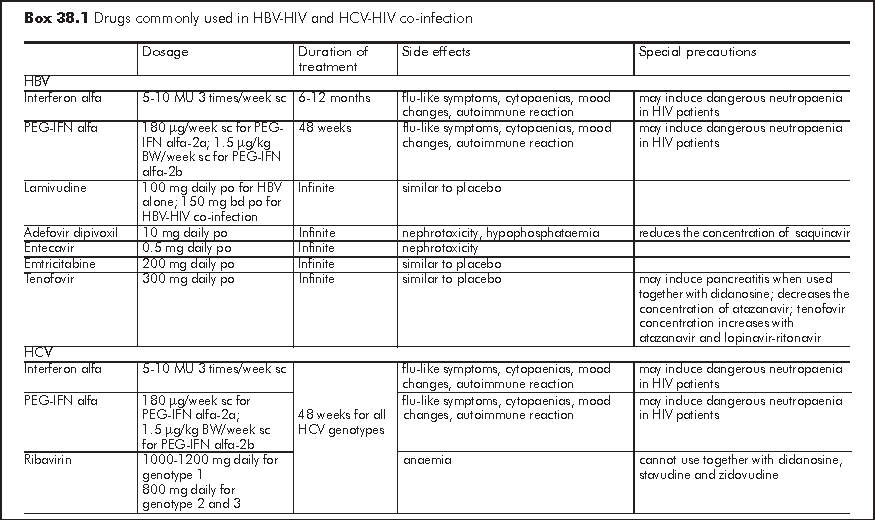
When there is the need to discontinue lamivudine, emtricitabine or tenofovir, flare has to be monitored clinically and biochemically, in particular for those with marginal liver reserve.20 Box 38.1 summarises the drugs for HBV treatment.
The rate of sustained virological response of chronic hepatitis C treatment by PEG-IFN and ribavirin is favoured by genotype 2 and 3 HCV, low viral load (<800,000 IU/mL), younger age (<40), higher ALT (>3 times upper limit of normal) and absence of liver cirrhosis. Among these factors, HCV genotype is the most important. Current practice guidelines suggest commencing treatment regardless of ALT level and fibrosis stage in patients infected with HCV genotypes 2 and 3 because of the high success rate. For patients infected with other genotypes, HCV RNA should be checked and liver biopsy may be considered to assess the severity of liver fibrosis. It is recommended to defer treatment if the fibrosis staging is low (F0-1). Because the treatment of chronic hepatitis C is not always successful and involves considerable side effects, a conjoined decision with the patient is preferred. Since PEG-IFN and ribavirin treatment is associated with neutropaenia, it is prudent to delay treatment until immune reconstitution by HAART for patients with severe immunodeficiency (CD4 count <200 cells/μL). In contrast to HIV negative patients, the current recommendation is that all HCV genotypes have to be treated with 48 weeks of PEG-IFN and ribavirin in coinfected patients if the response is favourable at 12 weeks post-treatment.
For HIV patients with acute hepatitis C, monotherapy with conventional interferon or PEG-IFN may be considered if HCV RNA cannot be spontaneously eliminated within 3 months. Intensive interferon treatment has been reported to clear HCV in up to 98% of cases with acute hepatitis C.21 Nevertheless; these regimes have not been tested in patients with HCV-HIV co-infection. Box 38.1 summarises the drugs for HCV treatment.

Most of the current treatment recommendations are based on data from chronic viral hepatitis patients without HIV co-infection. The treatment of HCV-HIV co-infection is more established by a few larger-scale randomised controlled trials. Most reports on antiviral therapy in HBV-HIV co-infection are small case series without control groups.
On the other hand, acute hepatitis B in an otherwise healthy adult does not warrant treatment. Acute hepatitis B is however, prone to become chronic in HIV-infected patients. The role of antiviral drugs in HIV-infected patients with acute hepatitis B has not been evaluated.
Compared to chronic hepatitis C, the treatment of chronic hepatitis B is limited by a lower efficacy and the necessity of life-long administration. At present, there have been no trials demonstrating superior efficacy of combination antiviral therapy to monotherapy alone. Further studies on how to optimise HBV treatment in mono-infected and HIV-HBV co-infected patients are earnestly warranted. This may result in not only enhanced treatment success but also lower rate of drug resistance, which is a particular problem in patients co-infected with HIV.
1. Lok AS, McMahon BJ; Practice Guidelines Committee, American Association for the Study of Liver Diseases (AASLD). Chronic hepatitis B: update of recommendations. Hepatology 2004;39:857-61.
2. Dienstag JL, McHutchison JG. American Gastroenterological Association medical position statement on the management of hepatitis C. Gastroenterology 2006;130:225-30.
3. Salmon D, Robain M, Rockstroh JK, Benhamou Y. Therapeutic management of hepatitis and HIV infection in co-infected patients: results of a survey performed before the 2005 Consensus Conference. J Hepatol 2006;44(1 Suppl):S2-5.
Bodsworth NJ, Cooper DA, Donovan B. The influence of human immunodeficiency virus type 1 infection on the development of the hepatitis B virus carrier state. J Infect Dis 1991;163:1138-40.
Chen CJ, Yang HI, Su J, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA 2006;295:65-73.
Yang HI, Lu SN, Liaw YF, et al. Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med 2002;347:168-74.
Thio CL, Seaberg EC, Skolasky R Jr, et al. HIV-1, hepatitis B virus, and risk of liver-related mortality in the Multicenter Cohort Study (MACS). Lancet 2002;360:1921-6.
Gandhi RT, Wurcel A, Lee H, et al. Isolated antibody to hepatitis B core antigen in human immunodeficiency virus type-1-infected individuals. Clin Infect Dis 2003;36:1602-5.
Alter MJ. Transmission of hepatitis C virus--route, dose, and titer. N Engl J Med 1994;330:784-6.
Puoti M, Bonacini M, Spinetti A, et al. Liver fibrosis progression is related to CD4 cell depletion in patients coinfected with hepatitis C virus and human immunodeficiency virus. J Infect Dis 2001;183:134-7.
Di Martino V, Thevenot T, Colin JF, et al. Influence of HIV infection on the response to interferon therapy and the long-term outcome of chronic hepatitis B. Gastroenterology 2002;123:1812-22.
Chan HL, Hui AY, Wong VW, Chim AM, Wong ML, Sung JJ. Long-term follow-up of peginterferon and lamivudine combination treatment in HBeAg-positive chronic hepatitis B. Hepatology 2005;41:1357-64.
Benhamou Y, Katlama C, Lunel F, et al. Effects of lamivudine on replication of hepatitis B virus in HIV-infected men. Ann Intern Med 1996;125:705-12.
Benhamou Y, Bochet M, Thibault V, et al. Long-term incidence of hepatitis B virus resistance to lamivudine in human immunodeficiency virus-infected patients. Hepatology 1999;30:1302-6.
Benhamou Y, Thibault V, Vig P, et al. Safety and efficacy of adefovir dipivoxil in patients infected with lamivudine-resistant hepatitis B and HIV-1. J Hepatol 2006;44:62-7.
Benhamou Y, Fleury H, Trimoulet P, et al. Anti-hepatitis B virus efficacy of tenofovir disoproxil fumarate in HIV-infected patients. Hepatology 2006;43:548-55.
van Bommel F, Wunsche T, Mauss S, et al. Comparison of adefovir and tenofovir in the treatment of lamivudine-resistant hepatitis B virus infection. Hepatology 2004;40:1421-5.
Chung RT, Andersen J, Volberding P, et al. Peginterferon Alfa-2a plus ribavirin versus interferon alfa-2a plus ribavirin for chronic hepatitis C in HIV-coinfected persons. N Engl J Med 2004;351:451-9.
Torriani FJ, Rodriguez-Torres M, Rockstroh JK, et al. Peginterferon Alfa-2a plus ribavirin for chronic hepatitis C virus infection in HIV-infected patients. N Engl J Med 2004;351:438-50.
Carrat F, Bani-Sadr F, Pol S, et al. Pegylated interferon alfa-2b vs standard interferon alfa-2b, plus ribavirin, for chronic hepatitis C in HIV-infected patients: a randomized controlled trial. JAMA 2004;292:2839-48.
Saves M, Vandentorren S, Daucourt V, et al. Severe hepatic cytolysis: incidence and risk factors in patients treated by antiretroviral combinations. Aquitaine Cohort, France, 1996-1998. Groupe dEpidemiologie Clinique de Sida en Aquitaine (GECSA). AIDS 1999;13:F115-21.
den Brinker M, Wit FW, Wertheim-van Dillen PM, et al. Hepatitis B and C virus co-infection and the risk for hepatotoxicity of highly active antiretroviral therapy in HIV-1 infection. AIDS 2000;14:2895-902.
US DHHS. Guidelines for the use of antiretroviral agents in HIV-infected adults and adolescents.
Jaeckel E, Cornberg M, Wedemeyer H, et al. Treatment of acute hepatitis C with interferon alfa-2b. N Engl J Med 2001;345:1452-7.